The Agencia Española de Medicamentos y Productos Sanitarios (AEMPS) has ordered the withdrawal of two products from the market.
This is a commonly used medicine, but marketed by two different companies: ibuprofen 400 mg. sold under the brands Pensavital and Mabo-Farma.
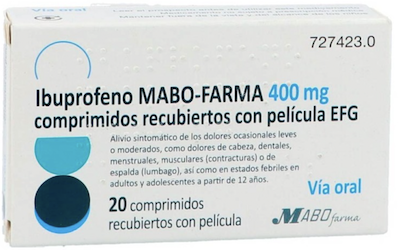
Information on the withdrawn products:
- Ibuprofen Mabo-Farma 400 mg coated tablets
- Box of Ibuprofen Mabo-Farma 400 mg coated tablets, withdrawn by the AEMPS.
- Batches and expiration dates: Batch: 468X, expiration date 07/31/2026; Batch: 469X, expiration date 07/31/2026; Batch: 470X, expiration date 07/31/2026
As reported by AEMPS, both medicines, produced by the company Farmalider S.A., presented results outside specifications in the dissolution test; i.e., how fast the pills dissolve.
For this reason, the agency has ordered the withdrawal from the market of all distributed units of the affected batches and their return to the laboratory through the usual channels.
The information from AEMPS indicates that this quality defect does not pose a life-threatening risk to the patient. Therefore, those who have it at home can continue using it as usual.
(News: Spain)
Keywords: Ibuprofen 400 mg, Pensavital, Mabo-Farma, Withdrawn from Market, AEMPS
